Bridge Device
- January 11, 2025
- Written by : Rebecca LaMarca, MSN, APN-BC, CNS
The Bridge from Masimo is the first FDA-approved medical device designed to help reduce opioid withdrawal symptoms.1
“Given the scope of the epidemic of opioid addiction, we need to find innovative new ways to help those currently addicted live lives of sobriety with the assistance of medically assisted treatment,” then-FDA commissioner Scott Gottlieb said in a press release at the time of approval in 2017.
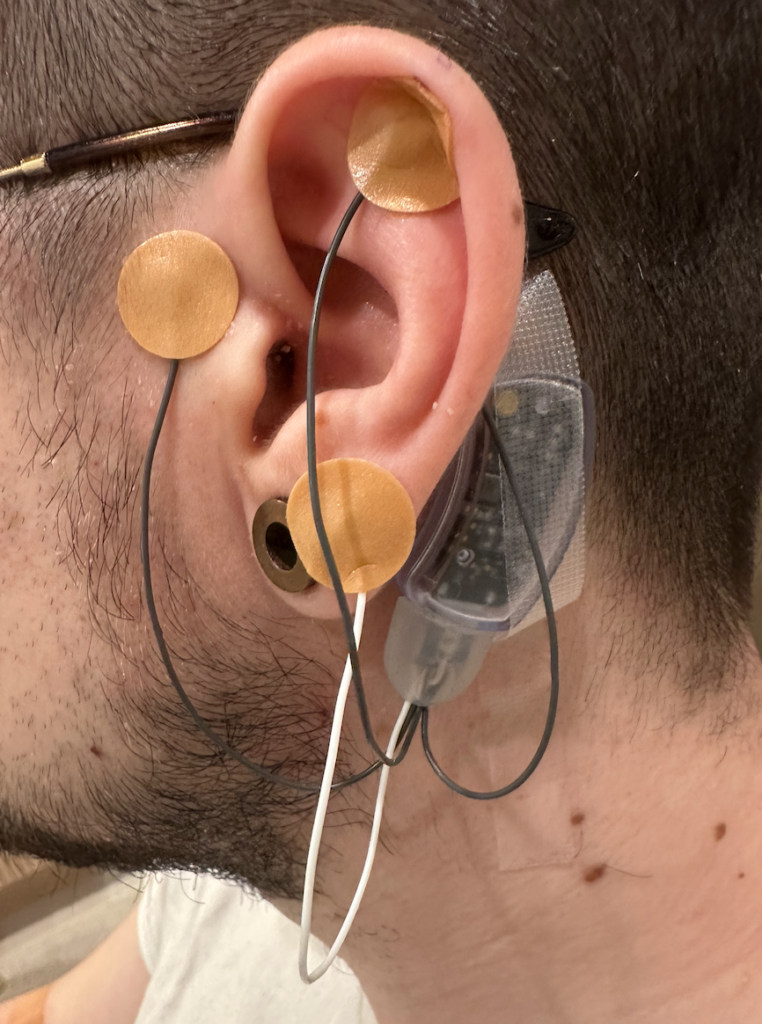
The small device, which needs to be applied by a qualified healthcare professional, contains a small battery pack and wires that get attached around the patient’s ear. The device produces small electrical currents that stimulate nerves, which transmit impulses to the brain and reduce withdrawal symptoms, providing relief for five continuous days.
According to Masimo, the Bridge can help with substance abuse detox in the following patients:
- Those starting medication-assisted treatment
- Those transitioning to antagonist treatment2
- Those tapering off of MAT, opioid therapy or opioid pain treatment when there’s dependence
Where Can I Get the Bridge Device?
The Bridge device for opiate withdrawal requires a prescription and is offered to healthcare professionals who undergo a training program to administer the device. IV Elements founder and nurse practitioner Rebecca LaMarca was certified to place the neuro-stim system device in 2019, becoming the only provider in New Jersey fully trained in placing the device.
The Bridge medical device is placed in about 15 to 30 minutes and left in place for a four- to five-day period and safely removed at the end of that timeframe. Make an appointment to have the Bridge device placed by IV Elements.
Bridge Device Concerns
While there were questions about how an early study that led to the Bridge’s FDA approval in a 2018 NPR report,3 a more recent 2023 study “demonstrated that NSS-2 BRIDGE™ can assist in opioid withdrawal and may increase initial treatment retention.”
That study included 16 participants, with eight participants in each group. One group received an active device, while the other received a “sham” device. The study evaluated the Clinical Opioid Withdrawal Scale (COWS) of each group.
Active group
- Mean COWS score at baseline: 17.125
- Mean COWS score after 30 minutes: 5.75
- Mean decrease: 11.375 (P<0.001)
Sham group
- Mean COWS score at baseline: 16.25
- Mean COWS score after 30 minutes: 12.875
- Mean decrease: 3.375 (P=0.186)
According to the researchers, “the active device group showed a statistically and clinically significant mean drop in COWS score compared to the sham group.”
REFERENCES
- Office of the Commissioner. (2017, November 15). FDA grants marketing authorization of the first device for use in helping to reduce the symptoms of opioid withdrawal. U.S. Food And Drug Administration. https://www.fda.gov/news-events/press-announcements/fda-grants-marketing-authorization-first-device-use-helping-reduce-symptoms-opioid-withdrawal
- Opioid antagonist. (2024, September 16). Cleveland Clinic. https://my.clevelandclinic.org/health/treatments/24878-opioid-antagonist
- Harper, J. (2018, May 2). Questions raised about study of device to ease opioid withdrawal. NPR. https://www.npr.org/sections/health-shots/2018/05/02/602908208/questions-raised-about-study-of-device-to-ease-opioid-withdrawal
Matta, C., & Wieseler, A. (2023). Prospective Study of Masimo NSS-2 BRIDGE; as a Non-Pharmacological Treatment for Acute Opioid Withdrawal Symptoms. Pain Management Nursing, 24(2), 238–239. https://doi.org/10.1016/j.pmn.2023.02.022
 Share
Share  Tweet
Tweet  Share
Share  Email
Email 


